 产品供求
产品供求
盐酸卡替洛尔
- 基本信息
- 物化性质
- 毒理性
- 结构与计算化学
- 下游产品
盐酸卡替洛尔 基本信息
- 中文名称:
- 盐酸卡替洛尔
- 中文别名:
- 盐酸卡替洛尔;
5-(3-特丁胺基-2-羟基)丙氧基-3,4-二氢喹诺酮盐酸盐
- 英文名称:
- 2(1H)-Quinolinone,5-[3-[(1,1-dimethylethyl)amino]-2-hydroxypropoxy]-3,4-dihydro-, hydrochloride(1:1)
- 英文别名:
- carteolol hydrochloride;
Carteolol monohydrochloride;
5-[3-(tert-butylamino)-2-hydroxypropoxy]-3,4-dihydro-1H-quinolin-2-one,hydrochloride;
Carteolol Hydrochloride
- CAS No.:
- 51781-21-6
- 分 子 式:
- C16H24N2O3.ClH
- 分 子 量:
- 328.88
- 精确分子量:
- 328.15500
- PSA:
- 70.59000
- EINECS:
- 257-415-7
- InChI:
- InChI=1/C16H24N2O3.ClH/c1-16(2,3)17-9-11(19)10-21-14-6-4-5-13-12(14)7-8-15(20)18-13;/h4-6,11,17,19H,7-10H2,1-3H3,(H,18,20);1H
- 安全术语:
- Poison by intravenous and intraperitoneal routes. Moderately toxic by ingestion and subcutaneous routes. An experimental teratogen. Experimental reproductive effects. When heated to decomposition it emits toxic fumes of NOx and HCl.
- 分子结构式:
-
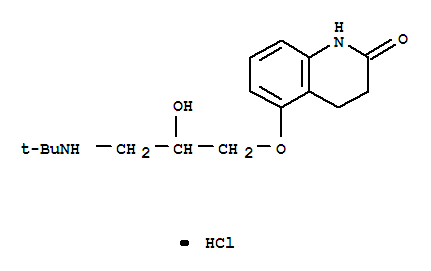
- SDS:
- 查看
盐酸卡替洛尔 物化性质
- 密度:
- 1.13g/cm3
- 熔点:
- 278ºC
- 沸点:
- 518.6ºC at 760mmHg
- 闪点:
- 267.4ºC
盐酸卡替洛尔 毒理性
CHEMICAL IDENTIFICATION
- RTECS NUMBER :
- VC8282000
- CHEMICAL NAME :
- 2(1H)-Quinolinone,
5-(3-((1,1-dimethylethyl)amino)-2-hydroxypropoxy)-3,4
-dihydro-, monohydrochloride
- CAS REGISTRY NUMBER :
- 51781-21-6
- LAST UPDATED :
- 199803
- DATA ITEMS CITED :
- 25
- MOLECULAR FORMULA :
- C16-H24-N2-O3.Cl-H
- MOLECULAR WEIGHT :
- 328.88
- WISWESSER LINE NOTATION :
- T66 BMVT&J GO1YQ1MX1&1&1 &GH
HEALTH HAZARD DATA
ACUTE TOXICITY DATA
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1330 mg/kg
- TOXIC EFFECTS :
- Behavioral - tremor
Behavioral - ataxia
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,159,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 390 mg/kg
- TOXIC EFFECTS :
- Sense Organs and Special Senses (Eye) - ptosis
Behavioral - convulsions or effect on seizure threshold
Lungs, Thorax, or Respiration - cyanosis
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,159,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 1950 mg/kg
- TOXIC EFFECTS :
- Behavioral - tremor
Behavioral - convulsions or effect on seizure threshold
Lungs, Thorax, or Respiration - dyspnea
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,159,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 50 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- USXXAM United States Patent Document. (U.S. Patent Office, Box 9,
Washington, DC 20231) Volume(issue)/page/year: #4535079
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 810 mg/kg
- TOXIC EFFECTS :
- Autonomic Nervous System - sympathomimetic
Vascular - measurement of regional blood flow
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 33,290,1983
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intraperitoneal
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 375 mg/kg
- TOXIC EFFECTS :
- Autonomic Nervous System - sympathomimetic
Vascular - measurement of regional blood flow
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 33,290,1983
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Subcutaneous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 600 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- IYKEDH Iyakuhin Kenkyu. Study of Medical Supplies. (Nippon Koteisho
Kyokai, 12-15, 2-chome, Shibuya, Shibuya-ku, Tokyo 150, Japan) V.1- 1970-
Volume(issue)/page/year: 12,668,1981
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - mouse
- DOSE/DURATION :
- 54500 ug/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- IYKEDH Iyakuhin Kenkyu. Study of Medical Supplies. (Nippon Koteisho
Kyokai, 12-15, 2-chome, Shibuya, Shibuya-ku, Tokyo 150, Japan) V.1- 1970-
Volume(issue)/page/year: 12,668,1981
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Mammal - dog
- DOSE/DURATION :
- 830 mg/kg
- TOXIC EFFECTS :
- Details of toxic effects not reported other than lethal dose value
- REFERENCE :
- ARZNAD Arzneimittel-Forschung. Drug Research. (Editio Cantor Verlag,
Postfach 1255, W-7960 Aulendorf, Fed. Rep. Ger.) V.1- 1951-
Volume(issue)/page/year: 33,290,1983
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 740 mg/kg
- TOXIC EFFECTS :
- Behavioral - tremor
Cardiac - pulse rate
Lungs, Thorax, or Respiration - dyspnea
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,165,1976
- TYPE OF TEST :
- LD50 - Lethal dose, 50 percent kill
- ROUTE OF EXPOSURE :
- Intravenous
- SPECIES OBSERVED :
- Rodent - rabbit
- DOSE/DURATION :
- 112 mg/kg
- TOXIC EFFECTS :
- Behavioral - tremor
Cardiac - pulse rate
Lungs, Thorax, or Respiration - dyspnea
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,165,1976
** OTHER MULTIPLE DOSE TOXICITY DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 12600 mg/kg/4W-C
- TOXIC EFFECTS :
- Liver - other changes
Endocrine - other changes
Related to Chronic Data - death
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,173,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 12600 mg/kg/12W-C
- TOXIC EFFECTS :
- Lungs, Thorax, or Respiration - changes in lung weight
Liver - changes in liver weight
Kidney, Ureter, Bladder - changes in bladder weight
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,173,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- SPECIES OBSERVED :
- Rodent - rat
- DOSE/DURATION :
- 102 gm/kg/26W-C
- TOXIC EFFECTS :
- Cardiac - other changes
Lungs, Thorax, or Respiration - other changes
Related to Chronic Data - changes in testicular weight
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 12,703,1976
** REPRODUCTIVE DATA **
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 21 mg/kg
- SEX/DURATION :
- female 9-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,221,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 700 ug/kg
- SEX/DURATION :
- female 9-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Embryo or Fetus - fetal death
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,197,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 7 mg/kg
- SEX/DURATION :
- female 9-15 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - urogenital system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,197,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 4350 mg/kg
- SEX/DURATION :
- female 15-22 day(s) after conception
lactating female 21 day(s) post-birth
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - eye/ear
Reproductive - Specific Developmental Abnormalities - musculoskeletal system
Reproductive - Effects on Newborn - growth statistics (e.g.%, reduced weight
gain)
- REFERENCE :
- JTSCDR Journal of Toxicological Sciences. (Japanese Soc. of Toxicological
Sciences, 4th Floor, Gakkai Center Bldg., 4-16, Yayoi 2-chome, Bunkyo-ku,
Tokyo 113, Japan) V.1- 1976- Volume(issue)/page/year: 4,59,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 24 mg/kg
- SEX/DURATION :
- female 15-22 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - stillbirth
- REFERENCE :
- JTSCDR Journal of Toxicological Sciences. (Japanese Soc. of Toxicological
Sciences, 4th Floor, Gakkai Center Bldg., 4-16, Yayoi 2-chome, Bunkyo-ku,
Tokyo 113, Japan) V.1- 1976- Volume(issue)/page/year: 4,59,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 525 mg/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,211,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 6975 mg/kg
- SEX/DURATION :
- male 73 day(s) pre-mating
female 2 week(s) pre-mating - 6 day(s) after
conception
- TOXIC EFFECTS :
- Reproductive - Fertility - post-implantation mortality (e.g. dead and/or
resorbed implants per total number of implants)
- REFERENCE :
- JTSCDR Journal of Toxicological Sciences. (Japanese Soc. of Toxicological
Sciences, 4th Floor, Gakkai Center Bldg., 4-16, Yayoi 2-chome, Bunkyo-ku,
Tokyo 113, Japan) V.1- 1976- Volume(issue)/page/year: 4,47,1979
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 700 ug/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,197,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Intravenous
- DOSE :
- 175 mg/kg
- SEX/DURATION :
- female 7-13 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Effects on Newborn - sex ratio
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,197,1976
- TYPE OF TEST :
- TDLo - Lowest published toxic dose
- ROUTE OF EXPOSURE :
- Oral
- DOSE :
- 36 mg/kg
- SEX/DURATION :
- female 7-18 day(s) after conception
- TOXIC EFFECTS :
- Reproductive - Specific Developmental Abnormalities - musculoskeletal system
- REFERENCE :
- OYYAA2 Oyo Yakuri. Pharmacometrics. (Oyo Yakuri Kenkyukai, CPO Box 180,
Sendai 980-91, Japan) V.1- 1967- Volume(issue)/page/year: 11,231,1976
盐酸卡替洛尔 分子结构与计算化学数据
计算化学数据
1.疏水参数计算参考值(XlogP):无
2.氢键供体数量:4
3.氢键受体数量:4
4.可旋转化学键数量:6
5.互变异构体数量:3
6.拓扑分子极性表面积:70.6
7.重原子数量:22
8.表面电荷:0
9.复杂度:354
10.同位素原子数量:0
11.确定原子立构中心数量:0
12.不确定原子立构中心数量:1
13.确定化学键立构中心数量:0
14.不确定化学键立构中心数量:0
15.共价键单元数量:2
山东默派生物科技有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:99
包装信息:多种包装规格kg
价格:洽谈
- 联系方式:
-
联系人:张经理
电话:13070690626
手机:13070690626
湖北云镁科技有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:99%
包装信息:335kg,25kg纸板桶/编织袋/塑料桶,10kg铝听/纸箱,1kg铝箔袋/氟化瓶
价格:1.0元 - 335.0元
- 联系方式:
-
联系人:方经理
电话:027-59206672
手机:18327059871
湖北魏氏化学试剂股份有限公司
- 产品介绍:
-
产品名称:卡替洛尔盐酸盐
Cas号:51781-21-6
纯度:99
包装信息:100g
价格:215.0元
- 联系方式:
-
联系人:刘涛
电话:027-59101766
手机:13125137661
上海吉至生化科技有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:99%
包装信息:100mgea,50mgea
价格:988.0元 - 1,538.0元
- 联系方式:
-
联系人:江珊
电话:021-57520831
手机:15316869680
宝鸡市国康海思化学技术有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:98
包装信息:1kg
价格:洽谈
- 联系方式:
-
联系人:杨经理
电话:0917-3909592
手机:13892490616
湖北鑫鸣泰化学有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:98%
包装信息:25/桶kg
价格:1.0元
- 联系方式:
-
联系人:黄经理
电话:027-51833411
手机:13237166274
武汉园星生物医药技术有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:
包装信息:100kg,200kg
价格:0.0元 - 100.0元
- 联系方式:
-
联系人:谢先生
电话:027-85558552
手机:15927172400
湖北魏氏化学试剂股份有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:98%
包装信息:25kg/桶
价格:洽谈
- 联系方式:
-
联系人:张经理
电话:027-59102966
手机:18717199209
湖北惠择普医药科技有限公司
- 产品介绍:
-
产品名称:盐酸卡替洛尔
Cas号:51781-21-6
纯度:99%
包装信息:100kg,200kg,300kg,400kg,500kg
价格:64.0元 - 100.0元
- 联系方式:
-
联系人:王浩
电话:19107121455
手机:19107121455
